FDA Approves Sickle Cell Gene Therapies; Federal Program to Aid Low-Income Access
March 19, 2024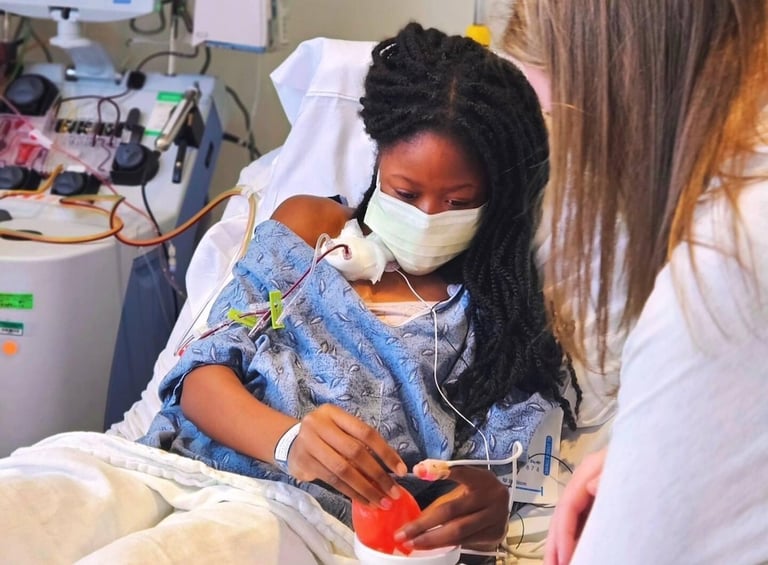
The U.S. FDA has approved new gene therapies for sickle cell disease.
A federal program is set to make these therapies accessible to low-income patients through Medicaid.
The program will negotiate drug discounts and is slated to begin next year.
Gene therapies have a high upfront cost, up to $3 million, but may save on long-term health care expenses.
Initial focus is on sickle cell treatments, with potential to expand to other conditions.
Sickle cell disease predominantly affects Black Americans, among others, with an estimated 100,000 affected in the U.S.
Illinois has committed to the program, while other states are in discussions, but some may struggle to participate without Medicaid expansion.
Summary based on 1 source
Get a daily email with more Science stories
Source
Missouri Independent • Mar 18, 2024
New way for states to cover pricey gene therapies will start with sickle cell disease • Missouri Independent