New Oligonucleotide RGLS8429 Emerges Safer, Effective for Kidney Disease, Avoids CNS Toxicity
November 29, 2025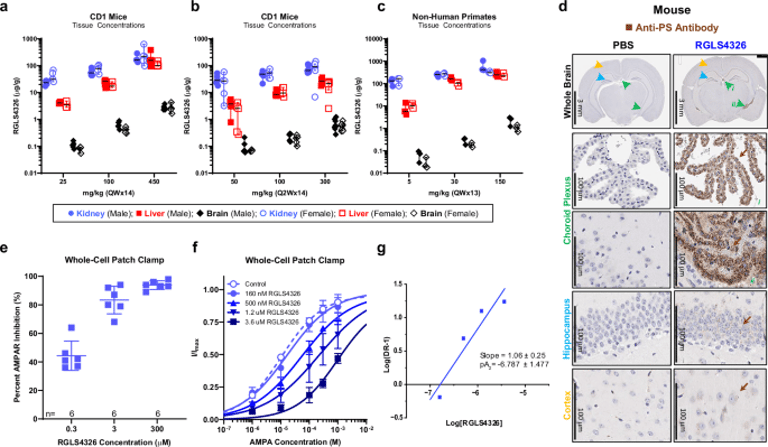
The first-generation anti-miR-17 oligonucleotide RGLS4326 caused CNS toxicity in nonclinical studies, which correlated with brain exposure and dosing, indicating off-target AMPA receptor inhibition rather than suppression of miR-17 as the primary cause.
A next-generation candidate, RGLS8429, showed no CNS toxicity in mice or chronic monkey studies at relevant doses and retained efficacy in reducing cyst growth while engaging miR-17 targets, supporting its advancement as a safer alternative.
The findings are discussed in the broader context of ON-related toxicities and structural biology efforts to map off-target interactions, underscoring the need for careful screening to avoid similar adverse effects in future candidates.
Replacing the 3' guanine with adenine in RGLS8429 abolishes AMPAR interaction and off-target toxicity while preserving anti-miR-17 activity, pharmacokinetics, and kidney-targeted efficacy similar to RGLS4326.
The study highlights the importance of SAR and off-target safety profiling for oligonucleotide therapeutics, identifying a mechanism by which a 3'G SNV drives CNS toxicity through AMPAR inhibition to guide future design choices.
The investigation builds on experience with the first-generation RGLS4326 for autosomal dominant polycystic kidney disease, noting CNS toxicity observed in chronic high-dose nonclinical studies in mice and monkeys.
Cryo-EM structural data show that the 3'-terminal guanine of RGLS4326 inserts into the AMPAR ligand-binding domain, stabilizing an open conformation that promotes channel inhibition and CNS toxicity.
Summary based on 1 source
