New Study Reveals Early Mutation Sequence Critical in Preventing Intestinal Tumors
December 5, 2025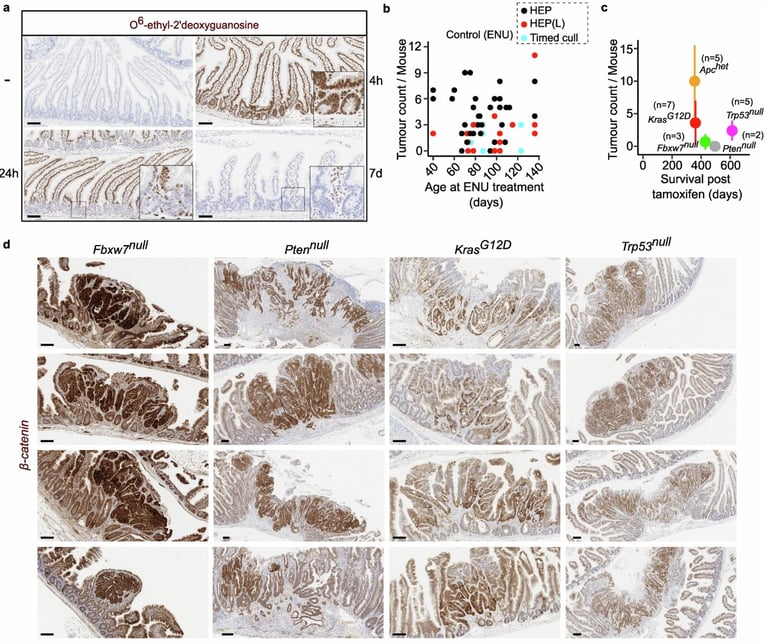
New research shows that the order in which driver mutations arise can shape cancer risk by trimming away vulnerable cells and enabling others to accumulate further alterations.
Mouse-model experiments reveal that strong negative selection eliminates most intestinal cells carrying tumor-initiating mutations, preventing cancer from developing.
Mutations don’t act in isolation; their effects depend on genetic context, and many cells with driver mutations are removed by negative selection unless supported by additional mutations.
This ongoing process of selection and environmental shifts shapes the landscape of intestinal transformation, guiding the path toward tumor formation.
The findings point to shifting prevention and treatment toward interrupting the earliest steps of tumor initiation and the tissue environment that governs mutational fitness, rather than focusing only on late, classic driver mutations.
Reference: Filipe C. Lourenço et al., Decay of driver mutations shapes the landscape of intestinal transformation, Nature (2025). DOI: 10.1038/s41586-025-09762-w.
Among surviving mutations, affected cells create a more permissive tissue milieu that raises the odds of further mutations occurring.
The work links early mutational events to later cancer evolution, underscoring the importance of mutation sequence in tumor development.
Some early changes, like certain APC mutations, reduce cell survival unless they follow other mutations, showing how initial genetic steps can shape tissue context for later drivers.
New mouse-model studies in Nature demonstrate that the sequence of cancer-driving mutations strongly influences whether intestinal tumors develop.
Researchers emphasize that initiating mutations may be negatively selected unless paired with other drivers, suggesting prevention could target early changes to cut cancer risk.
The research employed two approaches: introducing specific mutations first and then inducing random mutations, or vice versa, to probe the earliest steps of tumor formation.
Although many cells carry cancer-driving mutations, most are eliminated, with only a small minority influencing tumor development due to strong early negative selection.
The study, conducted by teams at the Wellcome Sanger Institute, Cancer Research UK Cambridge Institute, and the University of Glasgow, advances understanding of colorectal cancer risk with careful extrapolation from mouse data.
Summary based on 2 sources
Get a daily email with more Science stories
Sources
Medical Xpress • Dec 5, 2025
Order of cancer-driving mutations affects the chance of tumor development, study shows