New Study Unveils HIV Integrase's Structural Role, Paving the Way for Advanced Therapeutic Targets
February 18, 2026
A Nature study reveals that HIV-1 integrase forms an RNA-binding filament inside the capsid lumen, anchoring viral RNA to the core during maturation and implicating IN–CA interfaces in core assembly and RNA retention.
Led by Professor Juan R. Perilla at the University of Delaware, the study shows a new structural role for HIV integrase during early viral assembly, challenging the view that integrase acts only later.
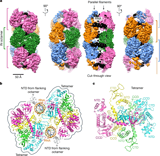
The research uncovers a previously unknown structural role for integrase inside the viral capsid during early maturation, where it forms filamentous structures linked to RNA and CA contacts.
Disruption of IN–LEDGF/p75 binding sites on the IN dimer interface overlaps with ALLINI targets, and disrupting this interaction reduces infection and increases eccentric particles, hinting at potential exclusivity with IN–CA interactions.
The work was enabled by longstanding collaborations among the Francis Crick Institute, Dana-Farber, Oxford, Birkbeck, Harwell, Imperial College London, and Harvard, with sustained support from NSF, NIH, and DOE.
The study stems from multidisciplinary collaboration across multiple institutions with public funding from major agencies, underscoring a broad, collaborative effort.
Previous research shows ALLINI inhibitors can disrupt integrase oligomerization and destabilize integrase–capsid interactions, suggesting a drug target beyond conventional replication inhibitors.
Functional validation shows mutations disrupting IN–CA contacts (CR1, CR2, CR4) or NTD stacking (e.g., V32 variants) severely impair infectivity and particle morphology, with compensatory mutations (IP6 interactions via K158) offering partial rescue.
Allosteric integrase inhibitors (ALLINIs) disrupt IN oligomerization and its interaction with the capsid and RNA, pointing to a therapeutic angle outside FDA-approved drugs.
Cryo-EM imaging required rapid freezing, stable environments, and extensive computation to build atomistic models of capsid–integrase interactions from millions of 2D images.
The IN octamer comprises two CTD-containing tetramers bound to RNA, using conserved positively charged residues to contact the RNA backbone, with NTD interactions linking octamer repeats.
Perilla emphasizes ongoing need for new HIV therapeutics and frames this discovery as a stepping stone toward novel inhibitors for clinical use.
Summary based on 3 sources
Get a daily email with more Science stories
Sources
Nature • Feb 18, 2026
Integrase anchors viral RNA to the HIV-1 capsid interior
EurekAlert! • Feb 18, 2026
Exposing a hidden anchor for HIV replication
BIOENGINEER.ORG • Feb 18, 2026
Revealing a Concealed Key to HIV Replication