Astrocytes Form Dynamic Networks in Mouse Brain, Potential Human Implications Explored
April 22, 2026
Experts unaffiliated with the study view this as a fundamental advance in nervous system structure, though the functional relevance remains to be clarified and could open new research avenues.
Funding for the study came from NIH grants and various foundations, with disclosures noting researchers’ affiliations and potential conflicts of interest.
The study, led by researchers at NYU Langone Health, was published in Nature on April 22, 2026.
While the findings are in mice, authors suggest potential relevance to humans and plan to explore how astrocyte networks relate to injury, disease, and aging in future work.
In vivo demonstrations showed that after injecting a regional tracer, administering biotin and using light-sheet imaging revealed infected astrocytes and neighboring in-network cells with biotinylated cargo, confirming gap-junction–mediated spread.
Double knockout of astrocyte gap junctions (Cx43 and Cx30) markedly reduces network size, indicating dependence on astrocyte gap junctions rather than vasculature.
Although networks exist in humans, it remains unknown whether the same regions are connected as in mice.
Researchers have revealed that astrocytes, long viewed as support cells, form extensive, long-range networks in the mouse brain that connect distant regions via gap junctions.
These astrocyte networks are dynamic and respond to sensory experience; for example, after trimming whiskers, the pathway from the sensory region contracts and rehangs with different astrocyte partners, indicating experience-dependent remodeling.
A custom tracing approach using virus-delivered network tracers and brain clearing mapped active astrocyte webs, uncovering pathways that can link areas not connected by neurons.
In vitro validation shows the fusion protein localizes to gap junctions, biotinylation occurs within the gap junction vestibule, and background is minimized by low native biotin in the CNS, aligning labeling radius with enzyme reach.
Quantitative mapping aligned with the Allen Brain Atlas showed largely distinct motor cortex networks, with more overlap in frontal/hypothalamic regions and some contralateral enrichment, suggesting a complex topology.
Summary based on 6 sources
Get a daily email with more Science stories
Sources
Neuroscience News • Apr 22, 2026
Astrocytes Build Long-Range Communication Networks in the Brain
NYU Langone News
Brain Support Cells Form Previously Unknown Network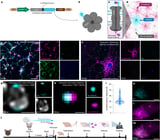
Nature • Apr 22, 2026
Astrocytes connect specific brain regions through plastic networks
Nature • Apr 22, 2026
Newfound brain network is a ‘secret system’ made of helper cells