FDA Approves First Oral IL-23 Blocker for Psoriasis: Icotyde Promises Convenience and Efficacy
March 18, 2026
The FDA has approved icotrokinra (Icotyde) as the first oral IL-23 receptor–blocking therapy for moderate-to-severe plaque psoriasis in adults and children 12 and older who weigh at least 40 kg and are candidates for systemic therapy or phototherapy.
Johnson & Johnson notes Icotyde was developed in partnership with Protagonist Therapeutics.
Pricing and availability details were not disclosed by the company in response to inquiries.
Icotyde is positioned as a convenient alternative to injectables like Skyrizi and Tremfya, with analysts forecasting rapid uptake and notable market share potential.
The current psoriasis treatment landscape usually begins with topical therapies and may progress to injectables or systemic treatments, with cost and convenience influencing decisions.
Industry and clinical experts highlight Icotyde’s potential for skin clearance with a favorable safety profile in a convenient once-daily pill form, aligning with calls from dermatology leaders to move beyond topical cycling to systemic therapy.
Wall Street analysts see blockbuster potential, with some estimates topping $8 billion in peak annual sales, while J&J projects over $5 billion across indications.
The development is situated within broader health-news coverage, referencing dermatology and health-outlet commentary.
Common side effects include headache, nausea, cough, fungal infection, and fatigue; serious infections are a risk due to immune modulation, with tuberculosis surveillance advised during and after treatment.
Accessibility and convenience are ongoing considerations driving Icotyde’s uptake in clinical practice.
In head-to-head trials, Icotyde achieved about 70% IGA 0/1 clearance and 55% PASI-90 by week 16, with adverse events similar to placebo through week 16 and no new safety signals by week 52.
The story is corroborated by multiple sources, including J&J releases, Reuters, and the American Journal of Managed Care.
Summary based on 5 sources
Get a daily email with more Science stories
Sources

Investing.com • Mar 18, 2026
US FDA approves J&J’s oral psoriasis pill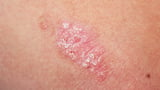
Medscape • Mar 18, 2026
FDA Approves Icotrokinra for Moderate-to-Severe Plaque Psoriasis
STAT • Mar 18, 2026
J&J wins approval for first-of-its-kind psoriasis pill
WSOC TV • Mar 18, 2026
FDA approves Johnson & Johnson’s oral pill for psoriasis, company says